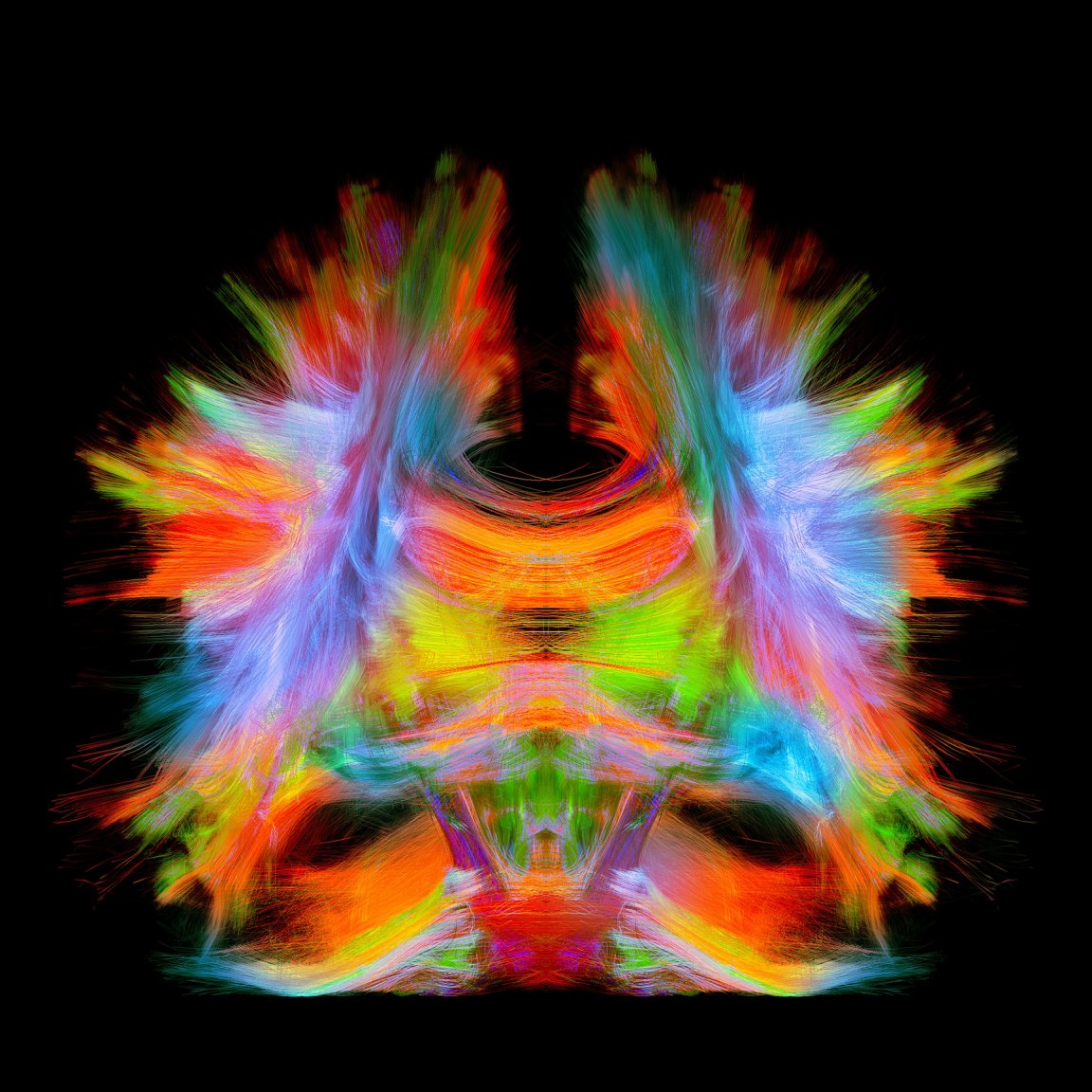
An Inside Look
If you’ve ever landed in the emergency room with a broken bone, fallen and hit your head, or been pregnant, then you already know how important medical imaging technology can be. If you’ve been diagnosed with appendicitis, a stroke, or any kind of tumor, those scans may have saved your life.
While medical imaging technology has given us the power to “see” inside the body, the tools of tomorrow will be more powerful still. The science behind this technology has evolved by leaps and bounds in the last few decades, but in order to apply those innovations to medical equipment, researchers need support.
If progress in this area has been slow, it hasn’t been for lack of ideas — it’s been a lack of funding. Now, there are signs that this might be changing, and researchers can already envision the implications of giving medical professionals better ways to see in the body. To do so would change more than the future of medicine: It would irrevocably change how we see ourselves.
Sight Unseen — Today
For centuries, the only way to see inside the body was through exploratory surgery, which has always come with risks — like an infection, hemorrhaging, and so on
Then, in 1895, Wilhelm Roentgen discovered X-rays, giving physicians a tool that could let them “see” the human skeleton without a single incision. And X-rays were only the beginning. Computed tomography (CT) scans take several X-rays and combine them to form a composite image, providing a more detailed internal view of organs, tissues, and bone. Positron-emission tomography (PET) scans identify changes at the cellular level: After a patient ingests a radioactive tracer, the scan tracks it as it moves through the body. This can be helpful not just for diagnosing a disease, but monitoring one that may progress, like metastatic cancer.
While these scans are essential to the practice of medicine, they aren’t without risk — even a simple X-ray exposes patients to a small dose of ionizing radiation, which can build up and pose health risks over time. So medical professionals do their best to use the scans sparingly.
In some cases, medical professionals can use other imaging techniques that don’t require any radiation at all. Ultrasounds, for example, use high-frequency sound waves to produce images of organs within the cavities of the body. They’re useful to help doctors diagnose conditions like ovarian cysts and gallstones. Inserting a catheter or gastrostomy tube becomes easier when the technician can guide them in via ultrasound. Of course, ultrasounds are most often associated with pregnancy, as they are used to construct real-time images of the fetus.
The downside, however, is that the images that ultrasounds produce aren’t particularly high-quality. The lack of detail means that, while an ultrasound might be able to detect a structure on or near the ovary, differentiating between a malignant tumor and a simple cyst usually requires imaging at a higher resolution or even a biopsy. The quality of the ultrasound image also varies depending on the skill of the sonographer, which could make the images less reliable, which could be a life-threatening issue.
While collectively these techniques have informed how doctors treat patients, there’s one technique that stands out above the rest: Magnetic Resonance Imaging (MRI). The MRI can provide a glimpse at the tissues and organs of the entire body, and it’s one of the best ways to take images of the brain.
First developed in the 1970s, MRIs use a combination of radio waves and magnetic fields to render images of our internal organs, soft tissues, bones, ligaments, and cartilage. The human body is full of proton-containing water molecules which will align when exposed to a magnetic field. They flip-flop at different rates depending on what tissues of the body they happen to be in. That’s how the MRI generates an image in which organs are (more or less) distinct from one another.
While MRIs are useful for finding abnormalities in blood flow within the brain, a close relative, functional magnetic resonance imaging (fMRI), can provide insights into the activity of cells in the brain.
And that, we suspect, is where many of the unsolved mysteries of the human body begin.
Much More to See
Research into medical imaging technology receives less attention than you might expect, even though patients benefit greatly (and very often their lives depend upon) this research. Less than two percent of grants from the National Institutes of Health are awarded to academic radiology departments, Richard Ehman, a diagnostic radiologist and director of the Mayo Clinic Advanced Medical Imaging Technology lab, told Futurism.
As it stands today, modern medical imaging methods are extremely useful for clinicians, so rather than devising new techniques, most researchers’ energies have been dedicated to refining existing techniques. In an interview with Nature, Roderic Pettigrew, the director of the National Institute of Biomedical Imaging said that today’s MRI machines are so specific that they can “track the diffusion of water molecules in the brain with such precision that you can compute their trajectories along fibre pathways,” giving physicians more information to better diagnose, monitor, and treat a number of neurological conditions.
The most cutting-edge MRI machines are found in research facilities, but that’s not what you’d find at your local hospital. As healthcare systems struggle under pressure to cut costs without compromising the quality of patient care, entire radiology departments languish and lag. Replacing an MRI machine is pricey, so it’s easy to understand why hospitals are using older models. Some US hospitals still use film.
“Most physicians would have a hard time imagining how they would take care of patients without these tools,” Ehman, of Mayo, said. While it’s unlikely that any Western medical practitioners lack the capability to take basic images of the body, complacency and a lack of research attention to new imaging techniques have hampered innovation. Stagnation is preventing us from seeing more.
And there is so much more to see.
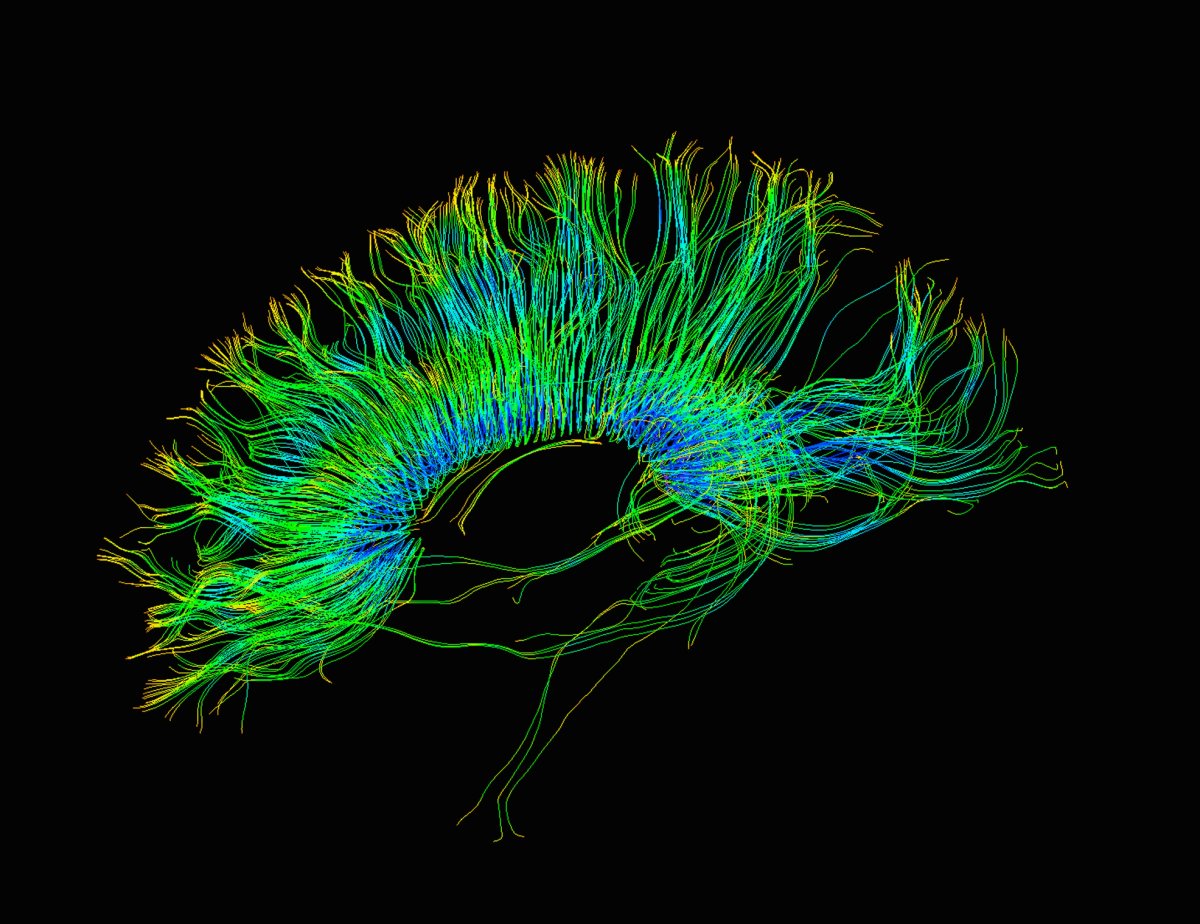
All the Better to See You With
Over the past 30 years, the design of MRIs has evolved to be faster, quieter, and more powerful — in large part because computer processors have gotten faster. Their evolution is far from over, and a team at the University of California Berkeley has set out to prove it. The team, composed of physicists and engineers, was recently awarded a $13.43 million BRAIN Initiative from the National Institutes of Health to develop the next generation of fMRI, dubbed the NexGen 7T, by 2019.
A MRI machine’s strength is measured in Teslas (T), which indicates the power of a magnetic field. When the magnetic field of our body (the tissues of which contain positively-charged water molecules) interacts with the magnetic field of the MRI, the tissue gives off a signal. The higher the power of the external magnet, the higher the intensity of that signal — and in turn, the higher the resolution of the images the MRI produces. Today, the world’s strongest MRI machines (which are generally isolated in research facilities and academic medical centers) are somewhere between 7T and 10T. Those that are most routinely used for patient care are much less powerful, clocking in at either 1.5T or 3T. Machines of that resolution are decently fast and accurate, good enough for most routine diagnoses: at 3T, an MRI can detect very small lesions in the brains of patients with multiple sclerosis, which a less powerful machine could miss.
But what if MRIs could allow us to go deeper, to see more — and to do so with breathtaking clarity? What could we glean about the progression of a neurodegenerative disease like multiple sclerosis (MS) if we could see nerves lose their protective myelin in real-time? If we could see the structures of the brain in greater detail, what could we learn about depression, anxiety, schizophrenia, or autism? If we could clearly see every nook and cranny, down to the axons and neurons, could observe the brains’ many processes as they happened, could we finally solve what some philosophers have called the “hard problem of consciousness?“
To inch us closer to doing so, researchers at UC Berkeley will need to achieve “a total redesign of nearly all of the [MRI] scanner components, not just an incremental change,” team lead David Feinberg said in the press release announcing the grant.
Another pair of researchers from UC Berkeley recently devised a way to make MRIs more safe and effective for children — a longstanding problem in the field. A physicist, electrical engineer, and a radiologist teamed up to create 3D-printed, flexible coils that fit onto garments. The resulting images were also remarkably crisp, which would benefit adults, too. Now the team is working with GE to further develop its design, possibly making up part of the next generation of MRI coils.
The applications for advanced imaging techniques go beyond our current uses. As Ehman noted, there are a number of measurements clinicians need to assess through the course of diagnosing and treating their patients. One example is that of measuring pressure within the human body. Getting these readings can require invasive, uncomfortable procedures, such as a spinal tap, that come with their own risks. As medical imaging once cut down the need for surgery, perhaps one day it could eliminate the need for needles, gauges, cuffs, and catheters.
Better imaging could also help us see elements of our own biology we never even knew existed. Invariably, more answers lead to more questions. And that will likely inform the path of medical research for decades to come.
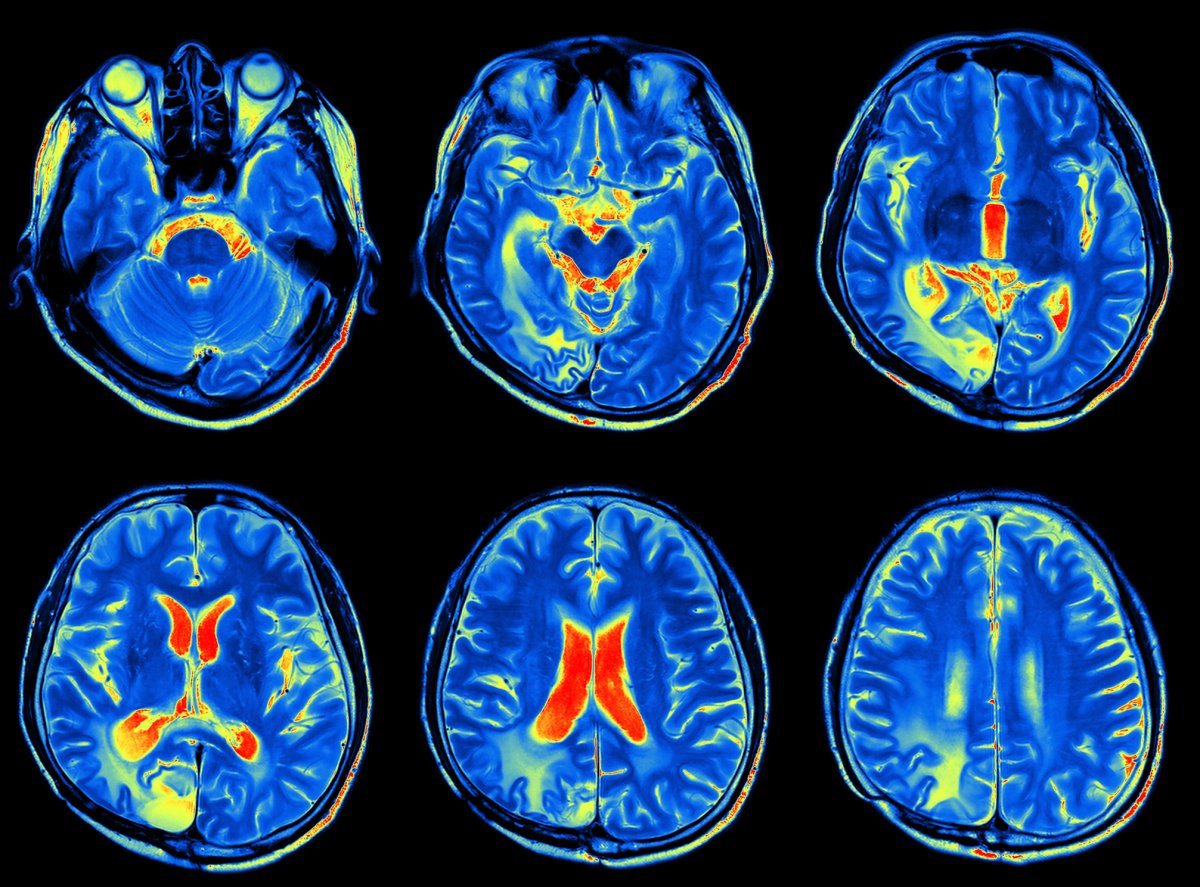
Other innovations in medical imaging may be a bit more dazzling to the average patient. At Massachusetts General Hospital, physicist Matthew Rosen is leading research into the clinical tools that are often paired with MRI machines, such as contrast dyes. His team is developing synthetic, biocompatible nanodiamonds that could replace contrast agents that are traditionally metal-based.
“Nanodiamonds have the potential to revolutionize the field of molecular imaging,” Rosen said in a press release, adding that their tiny size could enable radiologists to track any process at the cellular and subcellular level.
The team has only conducted lab-based tests and haven’t yet figured out how to best introduce the nanodiamonds into the human body (possible routes, as with any tracer, would include ingestion, infusion, or injection), according to a study Rosen and his team published in Nature Communications in March. There’s a long way to go before patients will have the option of taking a shot of medical-grade bling.
Technological progress made over the past century has transformed how we work, how we socialize, and how we date. The same goes for medicine, and for medical imaging in particular. That progress seems poised to continue — advanced computing, artificial intelligence, automation, and deep learning will improve medical technologies involved in everything from scans to surgery.
“I think that, in the future, we may look back at this time and it will seem that we were making very limited use of our best imaging technologies,” Ehman said (there are many complex reasons for this, he noted, including limited federal funding and the challenges of implementing new healthcare technologies). But Ehman remains hopeful that, despite these challenges, the future of medical imaging technology will transform medicine once again. Perhaps we can’t count on a renaissance, but having crystal clear vision will no doubt supply a few answers — and we should stay eager to be dazzled by them.
Disclaimer: the editor of this piece was a recipient of the Mayo Clinic’s 2017 journalist residency for surgery, which connected the author to sources quoted in this piece. The residency was paid for by the Mayo Clinic; however, neither the clinic nor any of its affiliates have editorial review privileges.