Killing Cancer with Apoptosis
Albert Einstein College of Medicine scientists have induced cancer cells to commit suicide with a new compound that leaves healthy cells untouched. They deployed their novel treatment approach against acute myeloid leukemia (AML) cells, which kill more than 10,000 Americans, and makes up about one-third of all new cases of leukemia, each year. Patients survive AML at a rate of only about 30 percent, making effective new treatments a hot commodity. And although the team has only tested the treatment on AML, it could have the potential to successfully attack other varieties of cancer cells.
“We’re hopeful that the targeted compounds we’re developing will prove more effective than current anti-cancer therapies by directly causing cancer cells to self-destruct,” associate professor of medicine and biochemistry and senior author Evripidis Gavathiotis said in a press release. “Ideally, our compounds would be combined with other treatments to kill cancer cells faster and more efficiently—and with fewer adverse effects, which are an all-too-common problem with standard chemotherapies.”
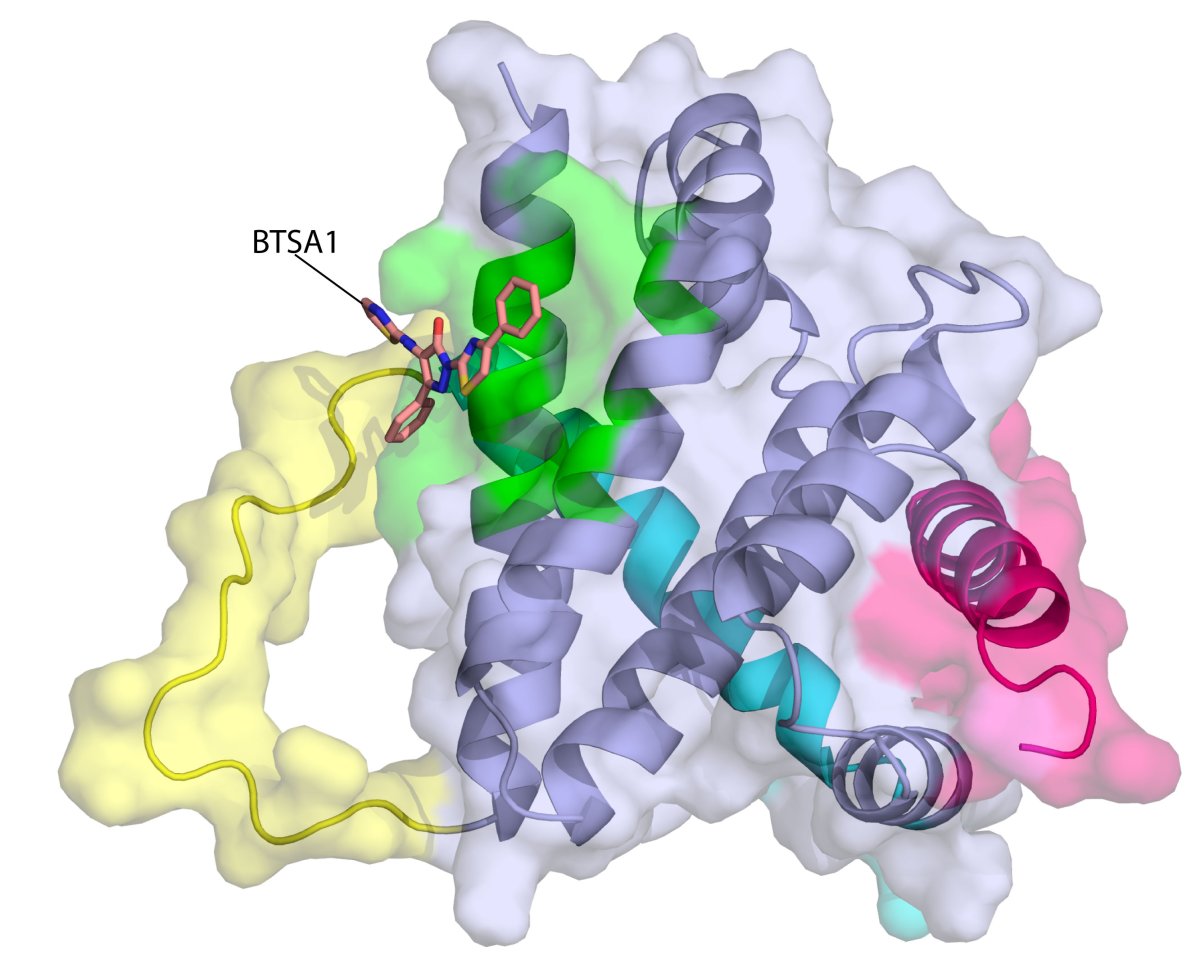
The new compound fights cancer by triggering apoptosis: a natural process the body uses to get rid of malfunctioning and unwanted cells. Apoptosis also takes place during embryonic development: trimming excess tissue from the growing embryo, for example. While certain existing chemotherapy drugs induce apoptosis indirectly by damaging the DNA in cancer cells, this treatment directly triggers the process intentionally by activating BAX, the “executioner protein.”
The Executioner Protein
Pro-apoptopic proteins activate BAX in cells. Once BAX molecules go to work, they find the mitochondria of target cells and drill lethal holes into them, scuttling their ability to produce energy. Cancer cells resist BAX and this process by producing large quantities of “anti-apoptotic” proteins that suppress BAX and even the proteins that activate it. The process discovered by these researchers wakes BAX up again and sends it back to work.
“Our novel compound revives suppressed BAX molecules in cancer cells by binding with high affinity to BAX’s activation site,” Dr. Gavathiotis said in the release. “BAX can then swing into action, killing cancer cells while leaving healthy cells unscathed.”
In 2008, Dr. Gavathiotis was part of the team that first described the BAX’s activation site’s shape and structure. Since that time, he has been searching for small molecules to activate BAX and produce sufficient activity to overpower the natural resistance cancer cells mount to apoptosis. His team screened more than one million compounds and narrowed the field to 500, many of them synthesized by the team, and then evaluated them. These results reveal the outcome of that search.
BTSA1 (short for BAX Trigger Site Activator 1) was the best compound against several different human AML cell lines, including those found in high-risk AML patients. BTSA1 was also able to induce apoptosis in AML cells without affecting healthy stem cells. In AML mice treated with the compound, there was a significantly longer survival rate: 43 percent of the control group was alive and AML-free after 60 days. The BTSA1-treated mice also exhibited no signs of toxicity.
“BTSA1 activates BAX and causes apoptosis in AML cells while sparing healthy cells and tissues—probably because the cancer cells are primed for apoptosis,” Dr. Gavathiotis said in the release. Next the team plans to test BTSA1 on other types of cancer using animal models.