Syringes, Shootings, and Stab Wounds
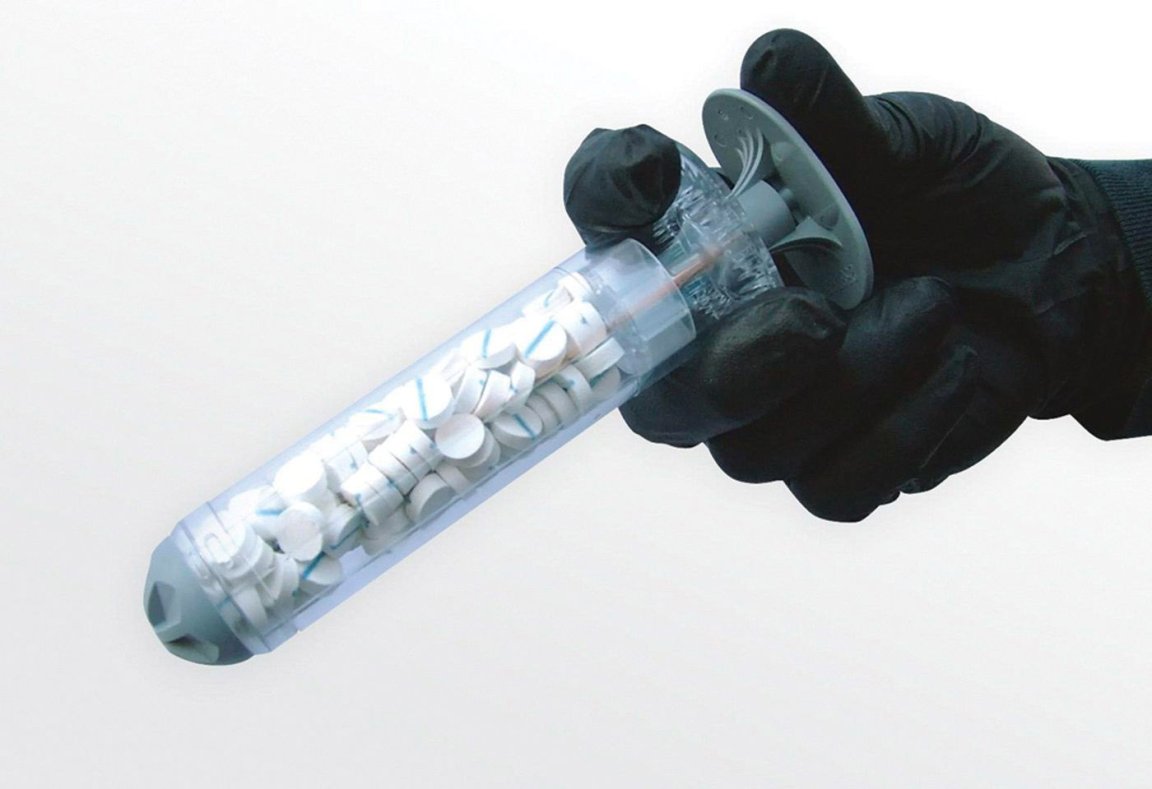
The U.S. Food and Drug Administration (FDA) has expanded their approval of the XStat 30, a sponge-filled syringe designed to treat gunshot and stab wounds, to include civilian use in the arm and leg.
The syringe design of the XStat 30 makes it capable of plugging deep and narrow wounds in a matter of seconds. Each of the devices contains small cellulose sponges that expand to up to 15 times their original size when brought into contact with blood.

These sponges can form a barrier to prevent blood from flowing out of the body by assisting with the clotting process and applying pressure to the wound. Each of the tiny sponges is outfitted with a small marker that can be detected via x-ray, which helps with surgical removal.
The manufacturer of the XStat 30, RevMedx, also received FDA approval to being marketing a smaller version of the device, the XStat 12, which is designed for use with smaller stab wounds and gunshots from smaller caliber weapons.
First Response
Though the XStat devices cost around $100 each and aren’t available without a prescription, they could be a major boon to first responders.
According to the United States Army Institute of Surgical Research, between 30 and 40 percent of all civilian deaths related to traumatic injuries are caused by blood loss. Furthermore, an estimated 56 percent of those instances occur before responders are able to get the patient to a medical facility.
The XStat 30 has been used on the battlefield since 2014 and in civilian hospitals since 2015, thanks in no small part to a $5 million grant from the U.S. Army that helped fund its initial development. However, prior to this new approval, the FDA only allowed civilian first responders to use the device in areas of the body where a gauze tourniquet isn’t an effective treatment, such as the groin and the armpit.
Extended FDA approval for these devices gives medical practitioners another tool for combating emergency situations, making them better equipped to save lives.