A Better Life for Patients
Good news for people with type 1 diabetes. The first “artificial pancreas” has been approved by the U.S. Food and Drug Administration (FDA).
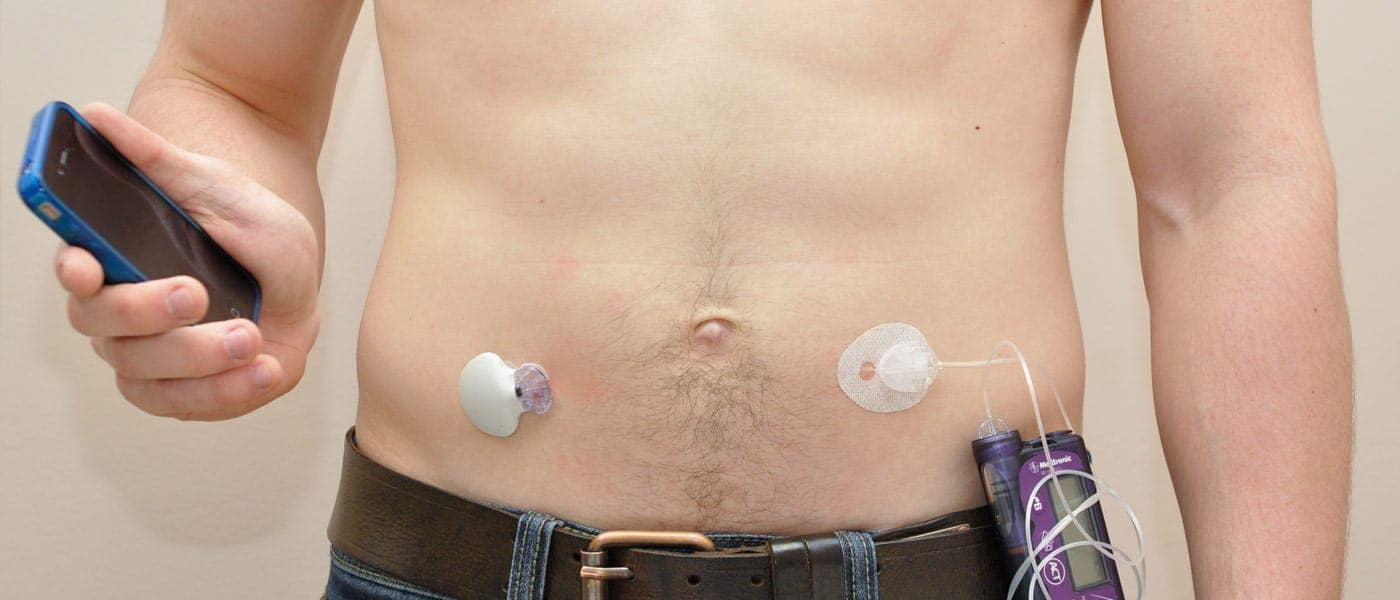
Innovative medical technology company Medtronic’s MiniMed 670G is the first device to combine an automated glucose monitor and an insulin pump. It administers an accurate amount of insulin as needed and will also automatically shut off insulin release when a drop in sugar levels is detected. This minimizes the risks of taking too much or too little insulin, both of which can have fatal consequences.
Medtronic describes the MiniMed 670G as “the first hybrid closed loop system in the world.” The device only requires patients to input mealtime carbohydrates and calibrate the sensors periodically, allowing them to live life more freely. It also helps them sleep well through the night and wake up with healthy glucose levels.
“This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin,” says FDA Center for Devices and Radiological Health director Dr. Jeffrey Shuren.
But even though the FDA has given the company the green light to send the device to the market for use by patients aged 14 and above, potential customers will have to wait just a little bit longer since Medtronic wants to conduct additional real-life testing before finally launching the MiniMed 670G in the spring of 2017. The company also aims to determine if the device would be safe for use in people below the FDA's age minimum.
Technology Saves Lives
In type 1 diabetes, the pancreas does not produce insulin, so the body loses its ability to naturally control blood glucose. If untreated, the disease can cause blindness, prompt kidney failure, or heighten risks for cardiovascular disease, among other complications, and in 2012, the American Diabetes Association estimated that 29.1 million Americans had diabetes. That's a whopping 9.3 percent of the population, and another 1.4 million Americans are diagnosed with the disease every year.
Currently, there is no cure for type 1 diabetes, but as medical technology advances, new hopes arise for people afflicted by this disease and so many others.
For example, a robotic sting ray imbued with live heart cells may one day give rise to a living, pumping human heart replacement, and familial cardiomyopathy patient Stan Larkin lived 555 days without a heart thanks to Syncardia, a technologically advanced portable blood-pumping device he carried in a backpack while waiting for a heart replacement. He even played basketball while using the device, showing that living without a vital organ is no reason for your court game to slip.
Share This Article