Coming Soon
The artificial pancreas has been getting a lot of buzz as the next big thing in managing diabetes, particularly Type 1 diabetes. In fact, it has recently entered clinical trials. Making them one step closer to home implementation.
A paper published in Diabetologia, the journal of the European Association for the Study of Diabetes, concluded that the artificial pancreas could enter the market as early as 2018.
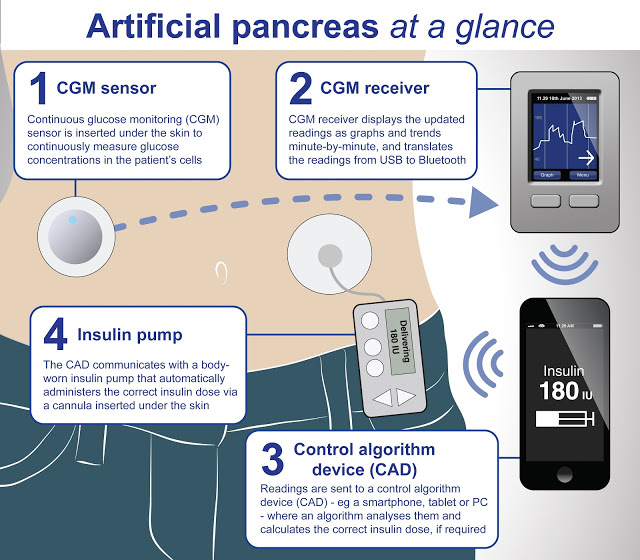
What is the artificial pancreas? The artificial pancreas is a device that monitors blood glucose in patients with type 1 diabetes and then automatically adjusts levels of insulin entering the body.
Currently, available technology allows insulin pumps to deliver insulin to people with diabetes after several readings with a glucose meter. But these are two separate components, and the task has been to integrate them into a device that does not need constant monitoring.
There are actually multiple organizations working toward the development of the technology. Some of these— Hovorka, Kovatchev and Phillip — have already partnered with companies to commercialize their efforts. Hovorka and Phillip are with Medtronic, Kovatchev is with a startup named TypeZero Technologies. Another, Damiano, founded a “public benefit” corporation called Beta Bionics last 2015.
Merging Technologies
The artificial pancreas is no mere amalgamation of the insulin pump and glucose meter. Rather, the artificial organ is able to adapt to improvements in diabetes technology over time. One of these improvements is in its glucose monitoring—essentially its ability to counteract the variability of insulin requirements between and within individuals.
Another improvement is the field of insulin analogues. Injected and other types of artificial insulin actually take some time to be effective, at times not fast enough. Fast-acting insulin analogues reach their peak 0.5 to 2 hours after injection, with their effects lasting 3 to 5 hours. The use of the even faster acting ‘insulin aspart’ analogue, as well as the development of other forms of insulin such as inhaled insulin is also under review for potential implementation.
The 2018 timeline actually include regulatory approvals, with reassuring attitudes of agencies such as the US FDA, which is currently reviewing one proposed artificial pancreas with approval possibly as soon as 2017.
The artificial pancreas and technologies like it are a stark reminder of how far we have come in terms of modern medicine, and where we will be going.