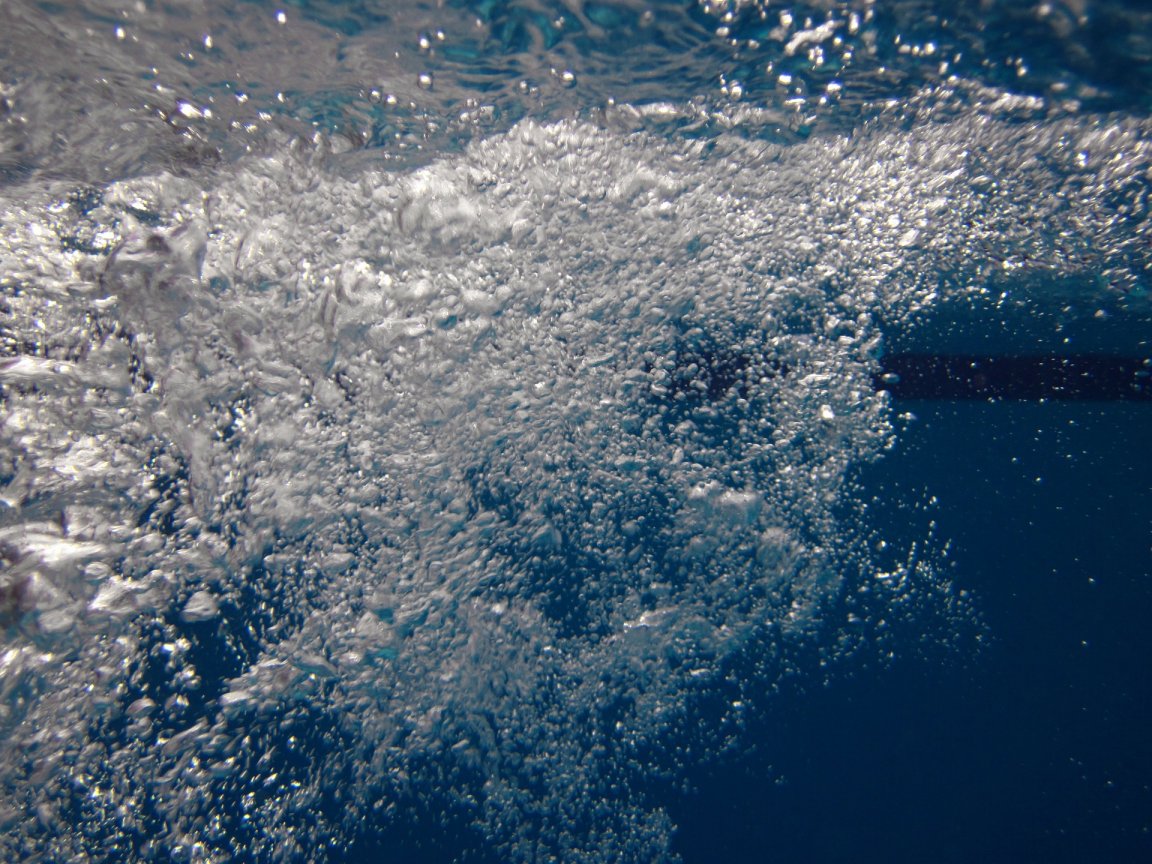
Taking Hydrogen from Seawater
Hydrogen fuel is yet another alternative form of energy than can be utilized to slowly wean the world away from fossil fuels — it’s a zero-emission form of energy, producing only water when burned. The problem, however, is that hydrogen gas (H2) for fuel is produced using a process known as steam methane, which also releases carbon dioxide (CO2), a key factor of global warming. The presence of CO2 lingering behind H2 makes it harder to facilitate its use over solar and wind energy.
Daniel Esposito, assistant professor of chemical engineering at Columbia Engineering, has been looking into a way to use water electrolysis — the process of separating water into hydrogen and oxygen gas (O2) — to produce hydrogen without carbon dioxide coming along for the ride. It took a bit of work, but Esposito and his team have developed a device that can do just that, and it may inspire the creation of larger devices capable of generating larger amounts of hydrogen fuel.
His creation is called a floating photovoltaic (PV) electrolyzer. As of now, it’s still a small lab-based prototype, but the final product would be developed into a complex floating on the ocean with an ironically similar appearance to a deep-sea oil rig. Instead of creating oil, though, this complex would produce H2 using sunlight and seawater.

Most commercial electrolyzers contain membranes that separate hydrogen and oxygen (O2) gases. Yet these membranes are susceptible to the impurities and microorganisms found within seawater, and can be damaged over time. Esposito’s device doesn’t use membranes, and therefore isn’t vulnerable to the same problems.
“The simplicity of our PV-electrolyzer architecture makes our design particularly attractive for its application to seawater electrolysis, thanks to its potential for low cost and higher durability compared to current devices that contain membranes,” said Esposito in a press release.
Fuel for the Real World
The new device relies on a unique configuration of electrodes, which separate and collect hydrogen and oxygen gas based on the buoyancy of their bubbles in water. This system doesn’t require pumping of the liquid within the device, and it produces hydrogen gas with 99 percent purity.

Esposito’s design isn’t ready for real operations just yet; it has only been tested on ideal electrolytes in the lab, and it isn’t prepared to tackle real seawater and the additional challenges it raises. Going forward, though, the team intends to improve their design to be more efficient, and also hopes to build bigger electrolyzers that can perform on a larger scale.
Given that other scientists have also been working on developing materials that can produce hydrogen fuel from seawater, we may have workable devices sooner than later.
“There are many possible technological solutions to achieve a sustainable energy future, but nobody knows exactly what specific technology or combination of technologies will be the best to pursue,” said Esposito in the press release. “Our challenge is to find scalable and economical technologies that convert sunlight into a useful form of energy that can also be stored for times when the sun is not shining.”