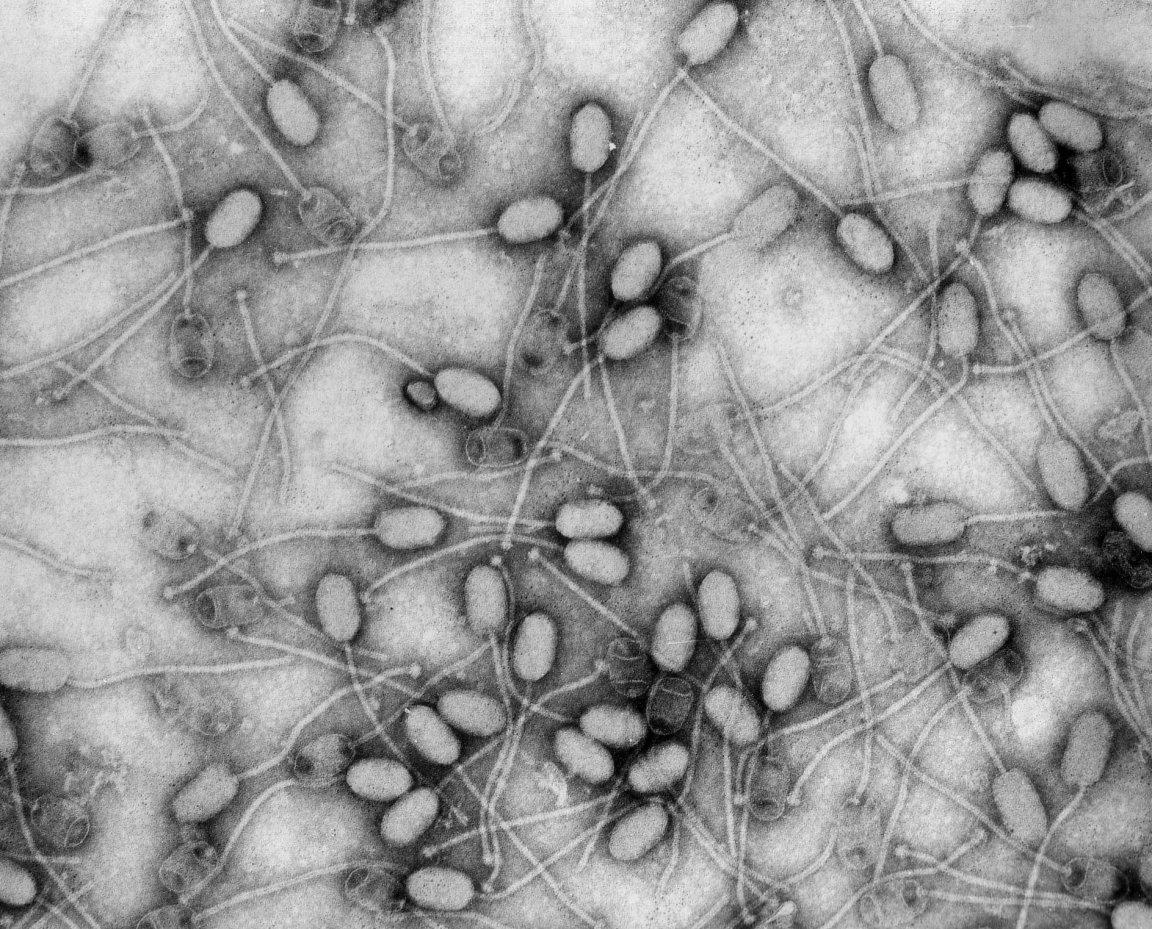
Going Viral
Several U.S.-based biotechnology companies are developing ways to harness the power of genetics as well as AI in the fight against antibiotic-resistant bacteria. Their ally? Viruses. More specifically, bacteriophages, usually just called phages.
We’ve been treating patients who have potentially life-threatening bacterial infections with these viruses for a long time, but typically only as a last resort. Phage therapy is imperfect in practice because it can be difficult to identify the bacteria, find the right treatment, and administer it before the patient succumbs. The potential of the process lies in whether it could be sped up without sacrificing accuracy or safety, which is exactly what many researchers are attempting to do.
The Resistance
Bacteria may be small, but they’re capable of wreaking great havoc on a population. The World Health Organization knows the potential of the pathogens should not be underestimated and has deemed antibiotic-resistant bacteria to be one of the most pressing global health concerns today.
For scientists, understanding how and why bacteria become resistant to the drugs we use to treat them is only the first step. In addition to the fact that the treatments we have today are becoming less effective, we also know that we just don’t have enough of them. And given the rate at which bacteria are becoming resistant to the limited options we currently have, global public health depends on these advances coming sooner rather than later.
Bacteria: Friend or Foe
The good news is, we know quite a bit about bacteria. The single-celled organisms have existed alongside (and inside) humans for the entirety of our shared history. Humans enjoy a great deal of symbiosis with the bacteria that live in our gut, for example. Much of the bacteria that live on our skin are more friend than foe; at the very least, they don’t harm us. Often they even help. It’s disease-causing bacteria that we have to protect ourselves against.

If you have a sore throat and fever, you might go to your doctor for a strep test. If it comes back positive for Streptococcus bacteria, you’ll be prescribed a course of antibiotics. These drugs either kill bacteria outright or make it difficult for them to continue to multiply. We’ve been using antibiotics in one form or another for a long time, and they have been very effective against a number of bacteria that sicken humans. The problem is, that’s changing. Because bacteria are changing — and they’re doing so faster than we can change the drugs in response.
Bacteria become resistant to drugs in a number of ways: sometimes by changing themselves to resist the effect of the antibiotic and survive it, and other times, by “neutralizing” the drug itself, rendering it ineffective. The more a bacterium gets exposed to a certain drug, the more opportunities it has to find a way to defend itself. It only takes a single bacterium figuring out how to survive an antibiotic for resistance to spread: when it divides and multiplies, it passes its survival strategy on.
Getting to the root of antibiotic resistance may well mean infiltrating the pathogens. To do that, scientists need something smaller, yet still powerful. That’s where viruses come in.
A Tech Boost
Advances in one field of science and technology often lend themselves to solving a problem in another. Techniques in genetic engineering and DNA sequencing, for example, have opened doors to life-saving treatments and feats of medicine that seem nearly miraculous.
Whether culling through data or assisting in surgery, robots and computers are partnering with humans to improve public health. Page therapy, which up until now has only achieved partial success, could be one of the main beneficiaries of this evolving partnership.
For example, the startup AmpliPhi Biosciences, is working to sequence the genome of disease-causing bacteria like Staphylococcus aureus so they can identify the best bacteriophages for the task of defeating them, assemble them into a treatment that would be ready-made and available to patients as soon as they need them.
Another startup, Adaptive Phage Therapeutics, is working on a machine learning algorithm that could process the genetic data of the bacteria and phage much more quickly than current methods (which take hours, if not days). Once the system is trained, it will be able to match the most effective phage to a particular bacteria.
“When a patient is critically ill, every minute is important,” Adaptive Phage Therapeutics CEO Greg Merril told MIT Technology Review. Time is of the essence not just for patients who are already sick, but to everyone around the world who is vulnerable.
Both AmpliPhi Biosciences and Adaptive Phage Therapeutics are planning clinical trials, which may even begin this year. As the threat of antibiotic-resistance grows, the results can’t come soon enough.