Teaching Old Bacteria New Tricks
Imagine training a dog or a horse to perform a new physical task, such as rolling over or jumping over a fence. Such a task may be possible within days. Now imagine training bacteria to perform a new physical task. Moreover, try training bacteria to perform something it was naturally genetically programmed to not do.
That is exactly what scientists from Berkley Lab did with Moorella thermoacetica.
There M. thermoacetica was coaxed to perform photosynthesis, even though it is naturally non-photosynthetic. What’s more impressive is that they even induced it to also synthesize semiconductor nanoparticles in a hybrid artificial photosynthesis system for converting sunlight into valuable chemical products.
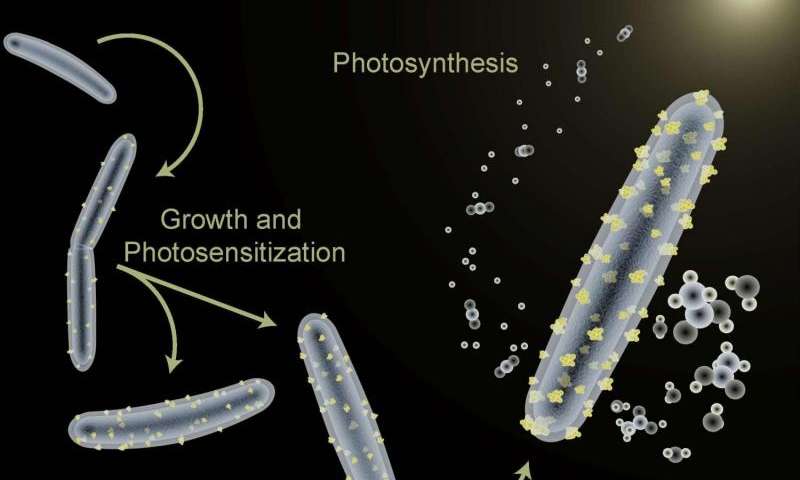
“We’ve demonstrated the first self-photosensitization of a non-photosynthetic bacterium, M. thermoacetica, with cadmium sulfide nanoparticles to produce acetic acid from carbon dioxide at efficiencies and yield that are comparable to or may even exceed the capabilities of natural photosynthesis,” says Peidong Yang, a chemist with Berkeley Lab’s Materials Sciences Division, who led the study.
The work was published as “Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production” in the journal Science.
Artificial Photosynthesis
If you aren’t already aware, photosynthesis is the process by which nature harvests sunlight and uses the solar energy to synthesize carbohydrates from carbon dioxide and water. Artificial versions of photosynthesis are already being explored.
Cadmium sulfide, on the other hand, is a semiconductor with a band structure. It is also well-suited for photosynthesis. As it is not only an “electrograph” (since it can undergo direct electron transfers from an electrode), but it is also an “acetogen,” as it can direct nearly 90% of its photosynthetic products towards acetic acid.
M. thermoacetica serves as the ideal model organism for demonstrating the capabilities of this hybrid artificial photosynthesis system. The study demonstrated that not only can biomaterials be of sufficient quality to carry out useful photochemistry, but that they may be even more advantageous in biological applications.