
Isaac Newton established that light is a particle. Many years later, James Clerk Maxwell made a theory that would disprove Newton–the theory of electromagnetism, which we still use today. In this theory, light is a wave. However, another great physicist came along and altered the theory again, once more asserting that light is a particle. This physicist was Albert Einstein. So it seems that we have a bit of a contradiction: We have everyone believing that light is a wave, based on the proven theories of Maxwell, but we also have two of the greatest physicists of all time theorizing light as a particle. So which is it?
Well, welcome to the world of quantum mechanics. Quantum mechanics says that light is both a particle and a wave. How is that possible? We don’t exactly know; we just know that it is true. This wave-particle duality does not only apply to photons of light, but also to all other sub-atomic particles.
Quantum mechanics also has an uncertainty principle which tells us that we can’t know for sure where a specific particle is at any given time, we can only know the probability of finding that particle at a specific point. The particle is literally at many places at once, and if we try to find the particle, nature will force the electron to choose where it will be based on the said probability. Einstein himself found this ridiculous, saying: “I like to think that the moon is always there, even when I’m not looking.” It also seems, in quantum mechanics, that making a measurement will somehow affect the thing that we are measuring. This is not because of our measuring techniques, it’s just that the particle seems to know when we are looking at it. Sounds a tad confusing, doesn’t it?
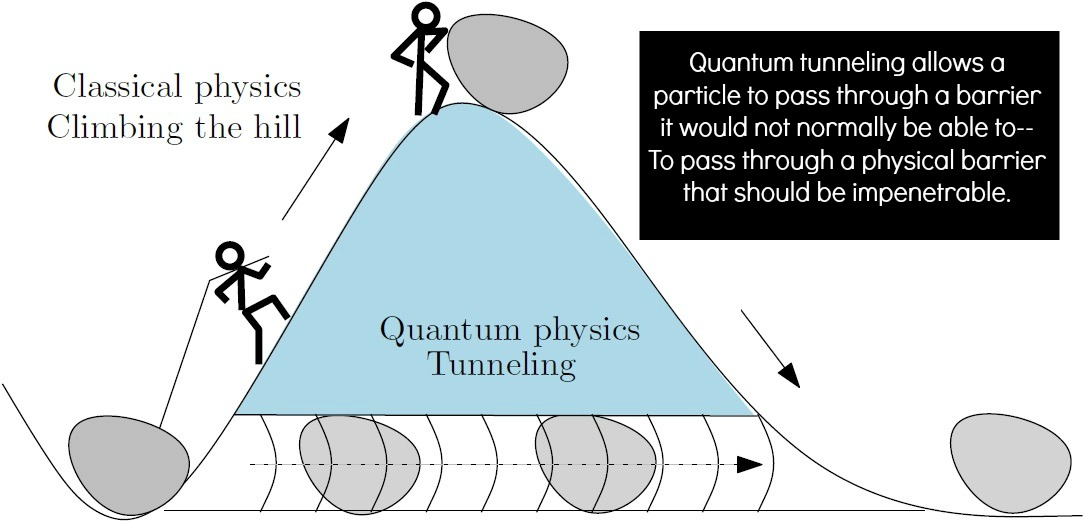
We all know teleportation in movies and video games. In Harry Potter, wizards can learn to “apparate” from one place to another. In the world of the very small, however, this is not merely science fiction, but an everyday reality. This phenomenon is called “quantum tunneling.” Electrons actually do it all the time inside the atom within a certain volume, called the electron cloud. Protons, on the other hand, also do this but within a much smaller volume because of the larger mass. Since we are made of particles that do this all the time, why can’t we do this ourselves? Why can’t we participate in quantum tunneling? Well, actually, we can, but quantum calculations only give us a very, very tiny possibility of doing so.

Probably the strangest mystery of quantum mechanics is on entangled particles. For example, in an atom, no two electrons can occupy the same quantum state. This means that 2 electrons in an atom, in the same orbital and shell, must occupy 2 distinct spin states. One would spin up, the other would spin down. When you change the spin of one electron, the other would immediately change to compensate to ensure that they do not have the same quantum state. The strange thing is, even if you put these two entangled electrons far apart, changing the spin of one electron will automatically change the spin of the other. And as it turns out, it doesn’t matter how far you put those two entangled particles apart, they can still affect each other instantaneously. Physicists even call this: “spooky action at a distance”. Physicists today still do not know how this happens.
Quantum mechanics reveals to us a very random and probabilistic world. This is very different from General Relativity’s picture of the universe, which is deterministic. Einstein did not like quantum mechanics at all, saying: “God does not play dice”, referring to the probabilistic nature of subatomic particles. But, whether Einstein liked it or not, quantum mechanics is true and has been proven by countless number of experiments.