The Weird World of Hydrogen
Hydrogen is the simplest element on the periodic table, and is the most abundant element in the known universe. It only has a single electron around a single proton, and yet, it behaves in fascinating ways under certain conditions.
Since it is a neutral electron spin-polarized gas, at absolute zero, hydrogen doesn’t form into a liquid, at around 14 K, unpolarized hydrogen will form stable molecules that can readily solidify.
More interestingly, over the past years, scientists have been experimenting with hydrogen and its isotopes in the hopes of finally unraveling the long-theorized form of the element, in which it dissociates into an atomic metal, possessing exotic properties at pressure levels in the millions.
The theories state that, at the metallic state of hydrogen, the element has high-temperature superconductivity, metastability, and at megabar pressures and low temperature, it forms in the liquid state. At such high pressures, solid hydrogen structure transforms into a graphene-like planes.
Revealing Metal
In a study recently submitted to Physical Review Letters, researchers from Harvard found new novel quantum phases of a hydrogen isotope, namely HD-IV* and HD-PRE, which are believed to be precursor transition states to the metallic hydrogen.
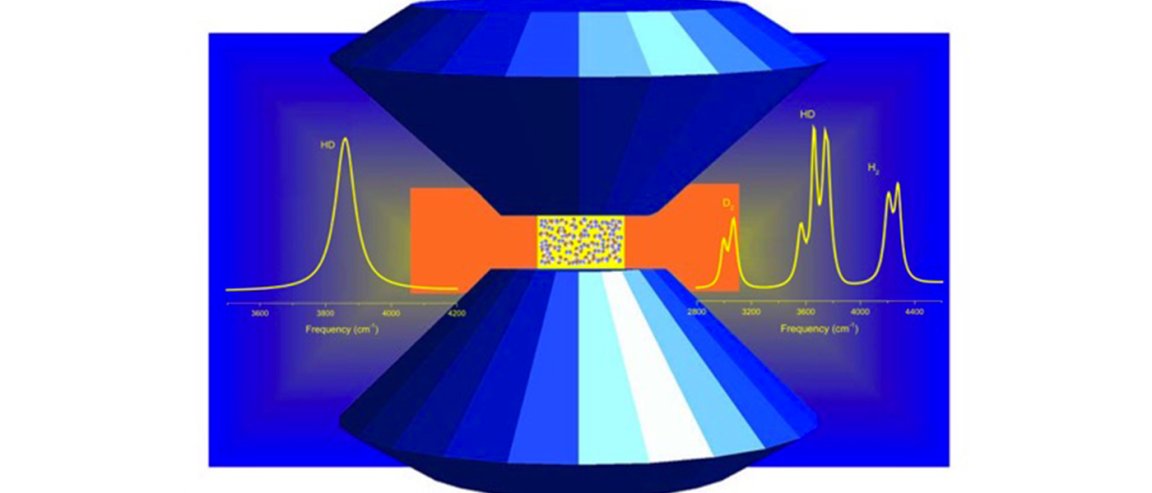
In this research, solid hydrogen deuteride (HD) was pressed with extreme pressure at 3.4 megabars, which is very very close to the pressure at the center of the Earth (3.6 megabars), using a diamond anvil cell.
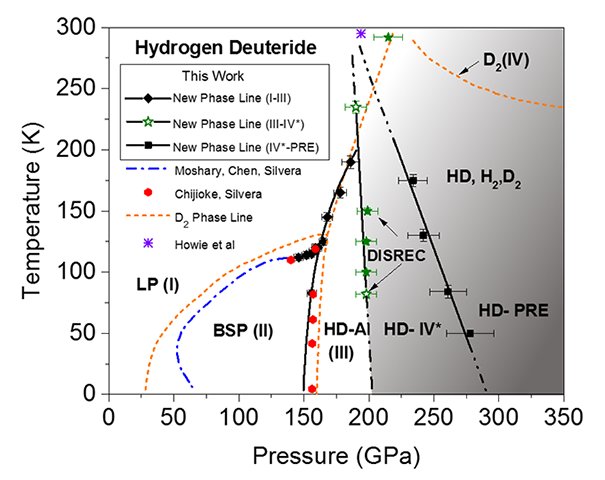
Results show that at 2 megabars, HD transitioned into HD-IV*, and with further increase of pressure, transformed into HD-PRE. At the HD-IV* transition state, the scientists observed a rapid dissociation of the molecule to recombine as the new isotopic solid, which they called DISREC.
Both states, however, still did not possess the sought-after metallic properties. Nonetheless, the scientists see this as another step towards the discovery of metallic hydrogen.