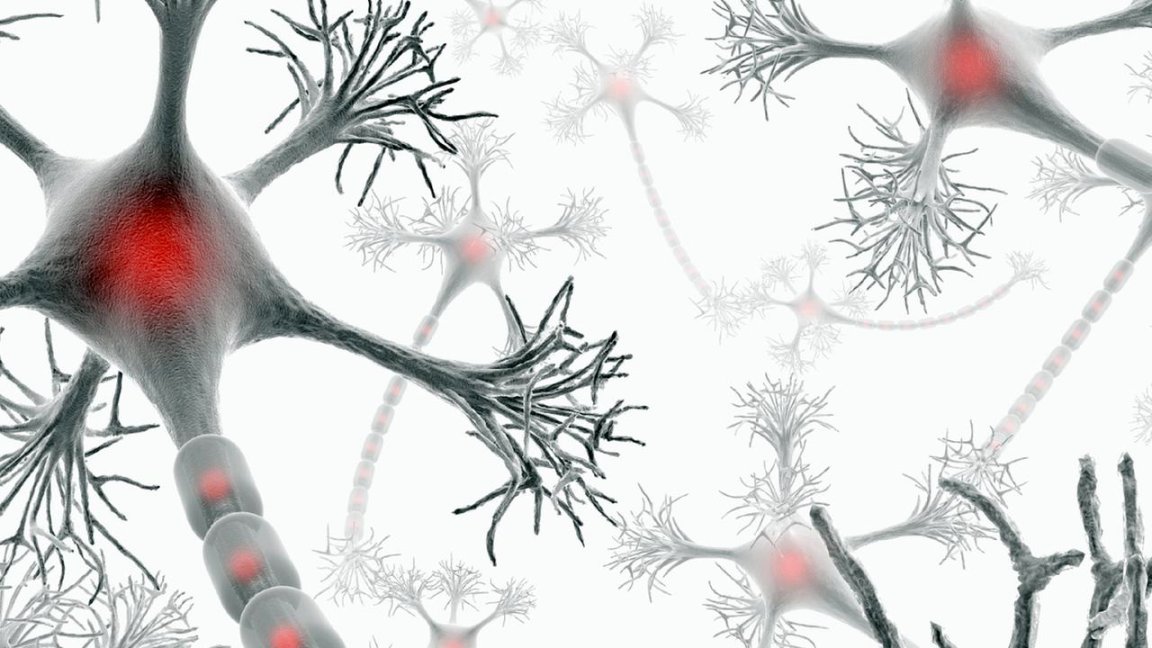
A Disabling Disease
Multiple sclerosis (MS) is an unpredictable and potentially disabling disease that cripples the central nervous system. It’s a widespread neurological condition that hits young adults, usually between the ages of 20 and 40, caused by an immune system disorder that mistakes a part of the brain as a hostile foreign object and attacks it. Though there are treatments available, particularly for its second state, multiple sclerosis remains incurable.
Progress is being made, however, with a new drug that can alter the immune system to slow damage to the brain for both states of MS. Ocrelizumab is the first known drug shown to work against the primary progressive form of MS. This primary progressive state is when MS gets worse, and being able to treat it is a “big news” in the eyes of doctors, as the BBC reports.
Promising Progress
The study, published in the New England Journal of Medicine, focuses on how ocrelizumab is able to destroy B cells, the part of the immune system that attacks the myelin sheath — the protective coating around nerves which serve as wire insulation for electrical signals to travel across nerves.
“The results shown by these studies have the potential to change how we approach treating both relapsing and primary progressive MS,” said Gavin Giovannoni, from Barts and The London School of Medicine and Dentistry, who was involved with the trials. “It’s very significant because this is the first time a phase three trial has been positive in primary progressive MS.”
The study notes that among 732 patients with progressive MS, those that showed deterioration dropped from 39 percent prior to ocrelizumab treatment to 33 percent with the new drug. Apart from these, patients who took the drug showed lesser brain loss detected on scans.
“It’s the first time a treatment has shown the potential to reduce disability progression for this type of MS, which offers a lot of hope for the future,” said Aisling McMahon, the head of clinical trials at the MS Society.