
Treatment Breakthrough
The autoimmune disease Alopecia areata (AA) is widespread, affecting an estimate of 4.6 million individuals in the United States, but developments in AA treatment have been slow. AA manifests as hair loss, and sometimes nail abnormalities, caused when the body’s immune system attacks hair follicles.
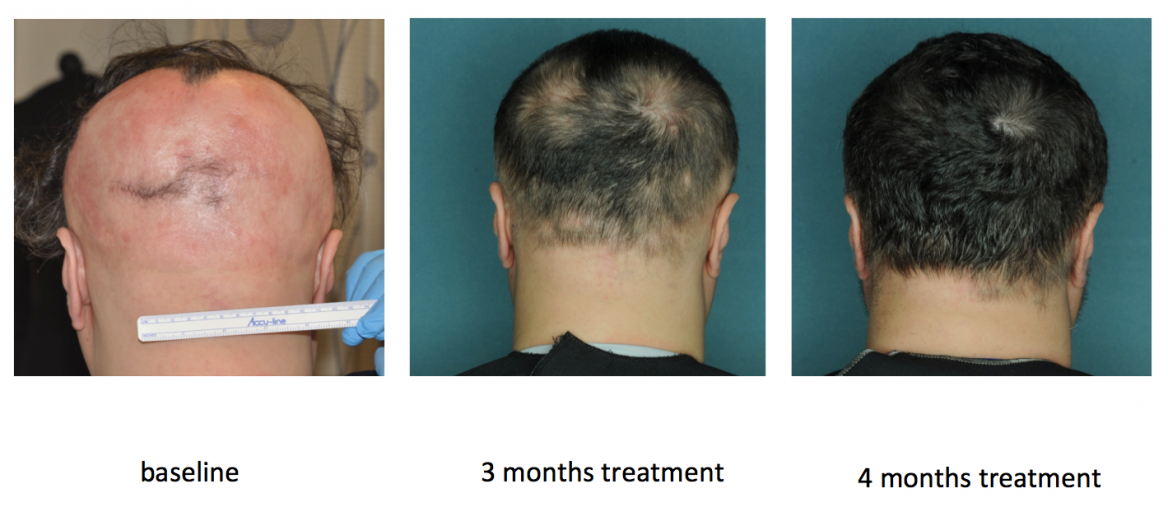
The first effective treatment for AA has been proposed by dermatologists from the Columbia University Medical Center (CUMC). Their study suggests that orally-administered ruxolitinib may be help treat patients with the disease.
An astounding result of 92 percent average hair regrowth among twelve patients with moderate to severe AA was obtained. The treatment had duration of three to six months, where the drug ruxolitinib was administered in 20 mg doses twice a day. Nine of twelve patients demonstrated a remarkable response, exhibiting hair regrowth of 50 percent or greater throughout the treatment.
A third of the patients had significant hair regrowth in the follow-up period after the medication stopped. The dermatologists report no serious adverse effects in the patients.
The drug administered, ruxolitinib, is a Janus kinase (JAK) inhibitor, which functions by reawakening the dormant hair follicles by blocking inflammatory signaling.
Encouraging Findings
“Although our study was small, it provides crucial evidence that JAK inhibitors may constitute the first effective treatment for people with alopecia areata,” said Julian Mackay-Wiggan, MD, MS, associate professor and director of the clinical research unit in dermatology at CUMC. “This is encouraging news for patients who are coping with the physical and emotional effects of this disfiguring autoimmune disease.”
The study from CUMC was funded by advocate organizations including Locks of Love Foundation and the Alopecia Areata Initiative. Continued research into treatment for AA bonds together a community that seeks to make breakthroughs in medicine to help more people get comfortable in their own skin.