TWO NEGATIVES MAKE A…?
Scientists have broken a 250-year-old rule of chemistry with the discovery of a new molecular structure. The study, which appears in the German scientific journal Angewandte Chemie International Edition, says researchers created a “supramolecule” born of a bond between two negatively charged molecules of bisulfate.
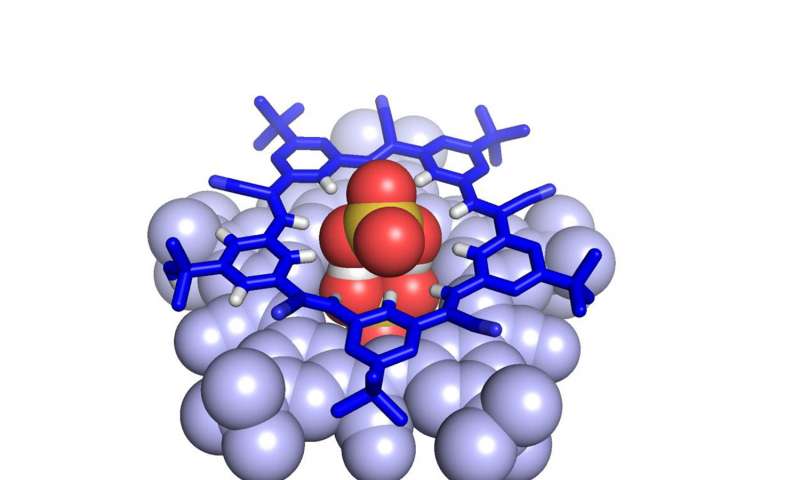
Until very recently, scientists argued how it’s impossible for negatively-charged molecules with hydrogen atoms attached — like bisulfate — to form viable chemical bonds.
“An anion-anion dimerization of bisulfate goes against simple expectations of Coulomb’s law,” said Indiana University professor Amar Flood, who is the senior author on the study, in a statement. “We believe the long-range repulsions between these anions are offset by short-range attractions.”
NUCLEAR POTENTIAL
The discovery of the “supramolecule” was made by binding bisulfates to cyanostar molecules: large, star-shaped molecules developed in Flood’s lab. These cyanostars can “grab” large, negatively-charged ions.
The molecule that defied a 250-year-old law now presents new possibilities that were previously unattainable, particularly in the search for chemical solutions to nuclear waste.
In nuclear waste treatment and conditioning, harmful compounds need to be converted to solids resistant to leaching in order to avoid water contamination. When these chemicals get into the water supply, they can trigger massive algae blooms that poisons water and kills fish in high numbers.
But the “supramolecule” is capable of removing sulfate ions from the nuclear waste. And it can also remove environmentally damaging phosphates from fertilizers.
“This paper is inspirational because it may launch a new approach to supramolecular ion recognition,” said Jonathan Sessler, chemistry professor at the University of Texas at Austin, in a statement. “I expect this will be the start of something new and important in the field.”