Non-Mythological Chimeras
The potential of stem cells for various treatments keeps scientists, like a team at the Salk Institute, to continually develop ways to produce human/animal (chimera) stem cells. Now, the chimera that these researchers are developing is not the multi-headed, fire-breathing monster of Greek mythology, but rather a genetic chimera. A genetic chimera is simply a singular organism made up of cells coming from different zygotes.
This team of Salk Institute researchers, led by Juan Carlos Izpisua Belmonte, are aiming to develop a chimera made of both human and pig stem cells. “The ultimate goal is to grow functional and transplantable tissue or organs, but we are far away from that,” said Izpisua Belmonte, researcher and author of the study published in the journal Cell. “This is an important first step.”
As expected, developing fully grown three-dimensional tissues and organs from stem cells in a petri dish wasn’t easy. “It’s like when you try to duplicate a key. The duplicate looks almost identical, but when you get home, it doesn’t open the door. There is something we are not doing right,” Izpisua Belmonte said. “We thought growing human cells in an animal would be much more fruitful. We still have many things to learn about the early development of cells.”
Izpisua Belmonte and Salk scientist Jun Wu’s experiment began with the production of a rat/mouse chimera, something that’s already been done by other scientists in 2010. They introduced rat cells into mouse embryos and allowed these to mature. They then proceeded to introduce human cells into an organism, opting for cows and pigs because their organs more closely resemble human organs in terms of size. They experimented with about 1,500 pig embryos over a four-year period.
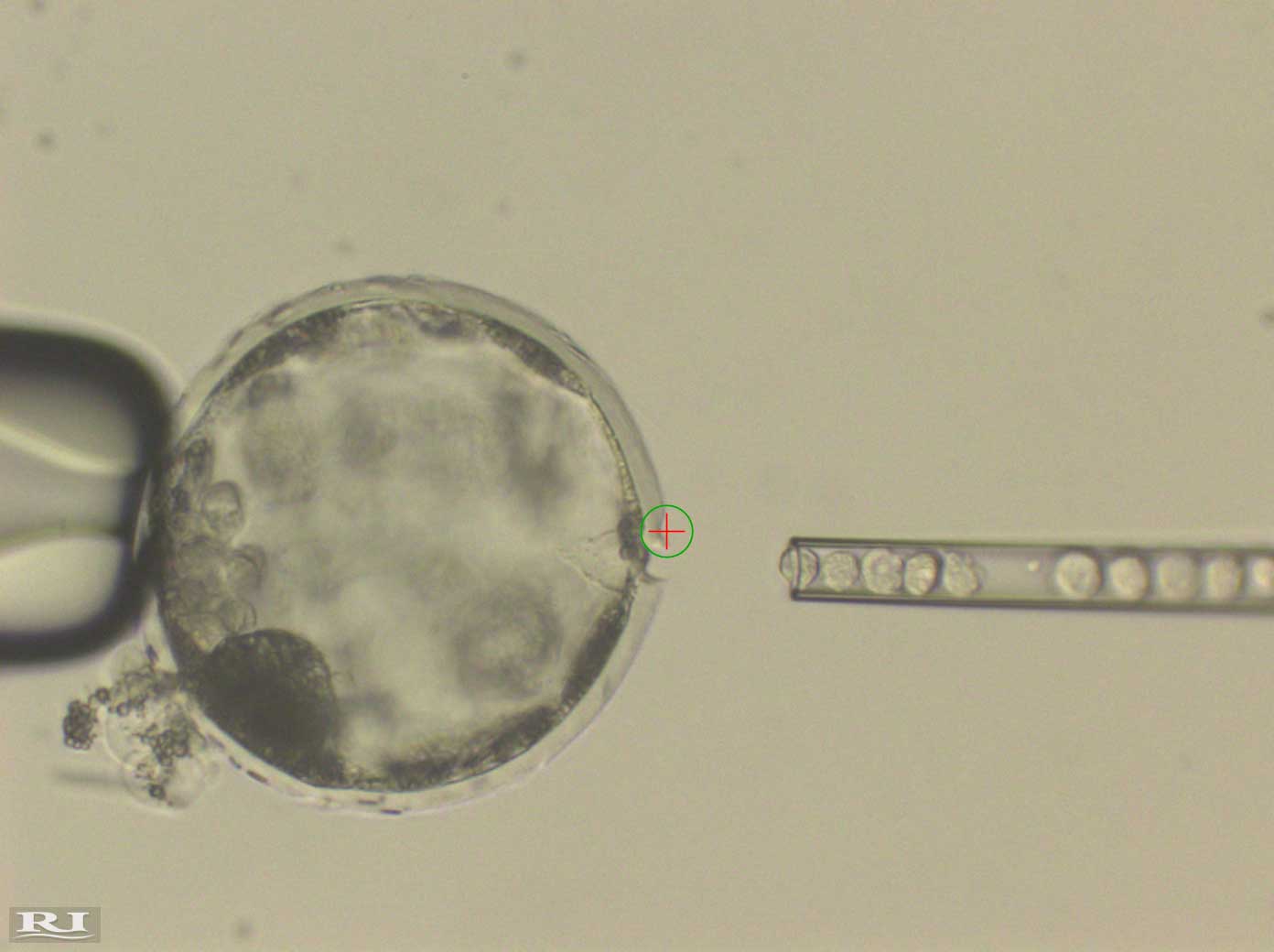
Exploring Cells
Several different forms of human stem cells were injected into pig embryos to determine which could best survive. Of these, those that showed a high survival rate and the most potential to continually develop were “intermediate” pluripotent stem cells. “Intermediate cells are somewhere in between” the two cell types, “naive” cells and “primed” cells, Wu explained. The former have unrestricted development and resemble cells in an earlier developmental origin. The later have already developed further but remain pluripotent.
The human cells that survived formed a human/pig chimera embryo. These were implanted into sows and were allowed to develop for three to four weeks. “This is long enough for us to try to understand how the human and pig cells mix together early on without raising ethical concerns about mature chimeric animals,” Izpisua Belmonte said.
However, even with the best-performing human stem cells, Wu stated that the level of contribution to the chimeric embryo was low. But this isn’t necessarily a bad thing. One concern about human/animal chimera embryos is that they would develop to be too human. “At this point, we wanted to know whether human cells can contribute at all to address the ‘yes or no’ question,” said Izpisua Belmonte. “Now that we know the answer is yes, our next challenge is to improve efficiency and guide the human cells into forming a particular organ in pigs.” The researchers are now relying on CRISPR to do this, editing pig genomes to open gaps that the human cells can fill in.
The goal of growing fully functional and transplantable tissue or organs is still far away. Thankfully, this team is not intending to create fully grown chimera organisms (the ethical issues of this “mad science” would be too difficult). For now, the researchers are happy that this study can help scientists understand how human stem cells grow and specialize. The human/pig chimera embryos could also give insights into early human development and disease onset.