Prize Winners
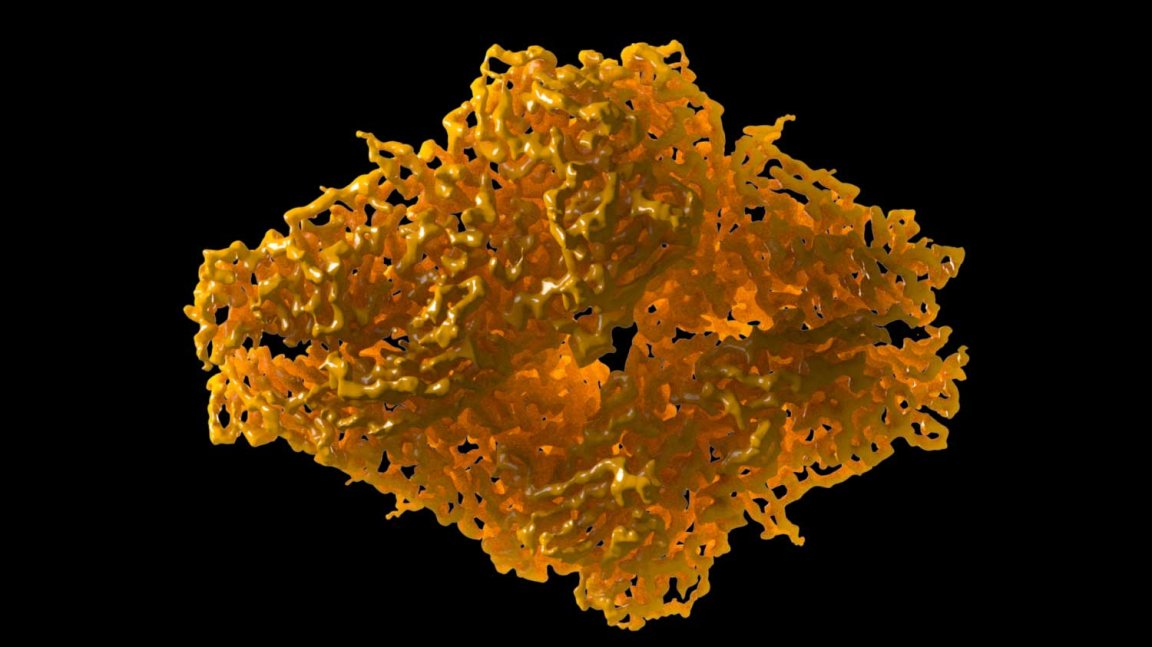
Jacques Dubochet, Joachim Frank, and Richard Henderson have been named as the recipients of the 2017 Nobel Prize in Chemistry, honoring their work on cryo-electron microscopy.
The technology has allowed researchers to create biochemical maps, which fill in gaps left by previous methods. Biochemicals can be frozen mid-movement, which allows previously unseen processes to be observed, offering up new insights into the chemistry of living organisms and aiding the development of new pharmaceuticals.

Scientists once assumed that electron microscopes couldn’t be used to inspect living cells, as their electron beams are capable of destroying biological material. However, in 1990 Henderson managed to successfully produce a 3D image of a protein at an atomic resolution.
Frank’s contribution to the technology was developed between 1975 and 1986 when he established a method of combining two fuzzy images produced by an electron microscope to produce one clear 3D model.
Meanwhile, Dubrochet was responsible for developing a method of adding water to the microscope’s vacuum and cooling it rapidly, helping biomolecules to retain their original shape.
A Closer Look
Since Henderson, Frank, and Dubrochet made breakthrough progress in the 1970s and 1980s, they’ve been hard at work ever since to perfect the process. It was only in 2013 that cryo-electron microscopy attained its desired atomic resolution.
The technology has allowed scientists to produce never-before-seen imagery of all different types of biomolecules, from the Zika virus to proteins that cause antibiotic resistance.
“[Cryo-electron microscopy] has opened up essentially a kind of new, previously unapproachable area of structural biology,” said Henderson in an interview with Nobel Media. “But the difference is there are quite a lot of structures in biology that were resistant, were recalcitrant to the other methods, like x-ray crystallography or nuclear magnetic resonance spectroscopy. So it has opened up essentially a kind of new, previously unapproachable area of structural biology.”