New Cancer Drug
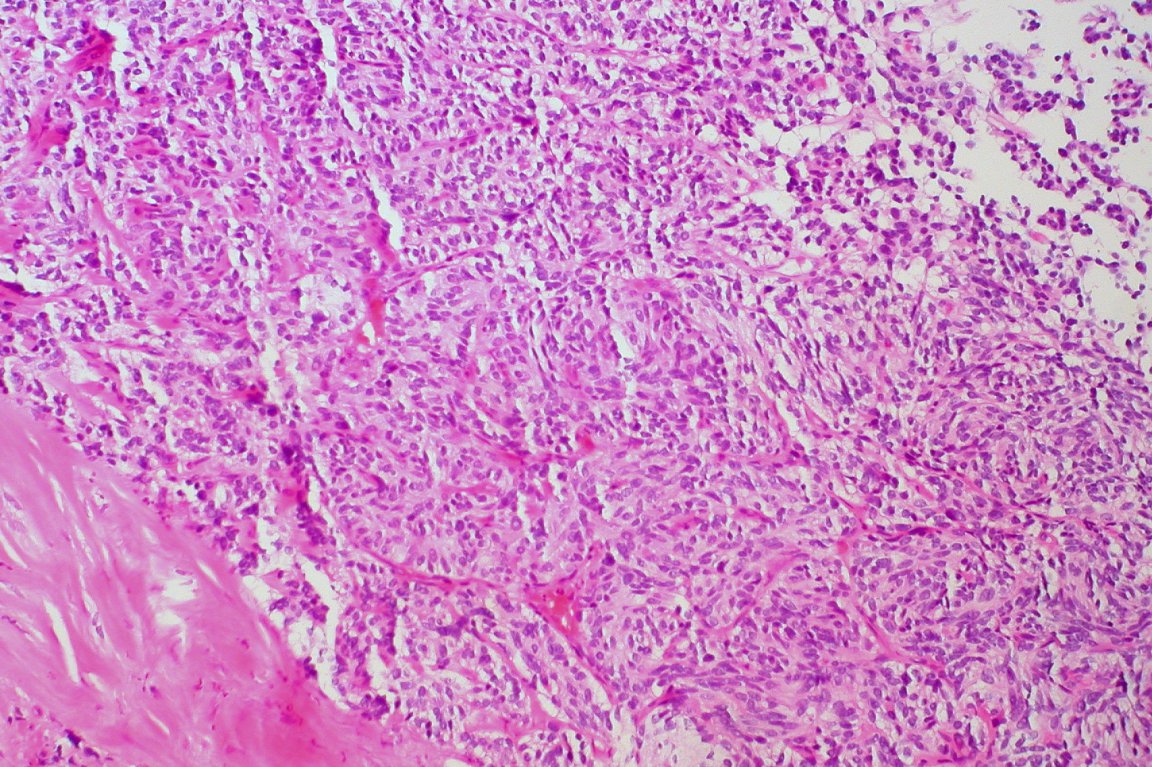
One new drug has doctors and pharmaceutical companies in a tizzy. Pembrolizumab (branded Keytruda) has recently been approved, in a hurry, by the Food and Drug Administration (FDA) to treat multiple tumors that arise from cancer in individuals with the same genetic abnormality.
During a clinical trail, the drug was tested in 86 patients. Of those who took part in the study, 66 patients had their tumors both significantly shrink and stabilize — meaning the tumors did not start to grow again. In 18 of these 66 patients — which is 21 percent of all patients — the tumors actually completely disappeared and not grown back whatsoever.
Now, this drug wouldn’t work for patients suffering from any type of cancer. For now, it is only approved to treat patients battling select varieties of advanced lung, melanoma, and bladder tumors.
Pros and Cons
While this is exciting progress, the treatment doesn’t come cheap. Just to test whether or not you might be a genetic match for the specific mutations that the drug targets costs between $300 and $600. The treatment itself currently costs $156,000 per year.
However, there is great hope that the drug itself and others that will follow in its footsteps will eventually come down in price, because this drug is truly the first of its kind. So what makes this drug so special? Well, it is the first approved drug in history that targets tumors from a specific, shared genetic profile, regardless of where the tumor is located.
Targeting tumors based off of genetic traits could help researchers and clinicians to more accurately target and treat cancers. Instead of just targeting the physical location of the tumor, treatments could be further tailored to the unique genetic profile of the individual patient. While this specific drug would only be effective for about four percent of cancer patients (though this would still help tens of thousands of patients), it could lead to a future where tumors are better targeted with genetic testing.