
A 94-Year-Old Invents Again
From powering electric cars to charging that smartphone you’re holding — the lithium-ion battery has grown to be one of the most commonly used batteries in the world. But one of its inventors, John Goodenough, has decided it’s time to upgrade.
Goodenough, who serves as a professor at the Cockrell School of Engineering of the University of Texas at Austin, has led a team of engineers to develop an entirely solid-state battery that could revolutionize how we use devices. The study behind the battery can be found in a paper recently published in the journal Energy & Environmental Science.
So what does this battery have to offer? The newly developed battery touts safety, faster-charging, and more recharge cycles. The battery can replace the current lithium-ion batteries found in handheld mobile devices, electric cars, and stationary energy storage. The new battery has three times the energy density of today’s batteries, meaning that it would give electric cars a greater range, recharge mobile devices in just minutes rather than hours, and would last much longer than its predecessor.
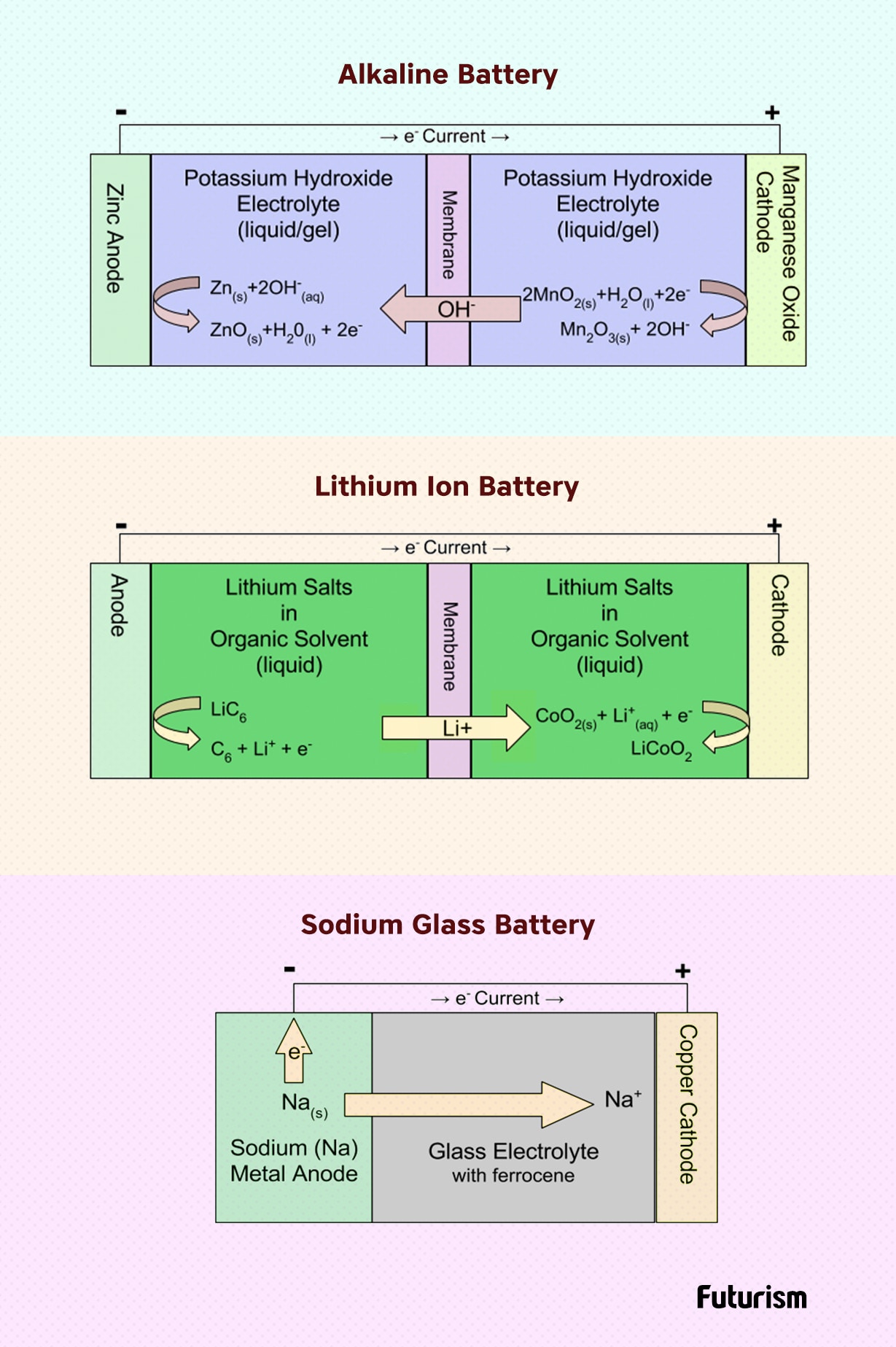
Unlike today’s batteries that use liquid electrolytes to transport the lithium ions, the new battery uses glass electrolytes instead. This reduces the risk of forming dendrites, whisker-like pieces of lithium that can grow within older models of batteries if they are charged too quickly, causing explosions and fires — similar to what we saw with Samsung Note 7.
Seawater: The Birthplace of Tomorrow’s Batteries?
The glass electrolytes opened up new possibilities for the team of inventors. Rather than relying on lithium-ions, Goodenough’s team was able to use a low-costing alternative — sodium.
With sodium ions readily available from sea salt, the team excited to design a new generation of batteries can be produced with earth-friendly materials, making the battery cheaper and, by extension, more accessible. Goodenough is currently working with battery makers to test the team’s work further, but he is confident in the work.