Conquering Cancer
According to the American Cancer Society, there are expected to be about 77,000 new cases of bladder cancer in the U.S. this year and 16,000 deaths. Fortunately, we are fighting back, and there are new weapons in our arsenal.
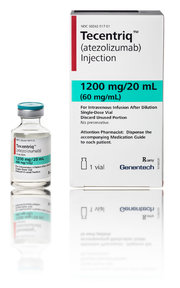
Just yesterday, the Food and Drug Administration approved a new immunotherapy drug developed by Roche to treat bladder cancer. The drug, called Tecentriq (known generically as atezolizumab), works by unleashing the body’s immune system to attack tumors.
To break this down, tumors and certain immune system cells can make a protein called PD-L1. It binds to a protein on the immune system T cells called PD-1, which then acts as a molecular brake, preventing the T cells from attacking the cancer cells.
Tecentriq, prevents the two proteins from binding, and that frees the immune system to attack. Roche argues that this will make its drug potentially safer, yet some experts see little difference among the drugs.
Roche’s study found that the drug was most likely to be successful for the roughly one-third of patients with high levels of PD-L1 near the tumor. 26 percent of those patients experienced significant tumor shrinkage, and 12 percent had complete remissions.
Longer Lives at a Cost
Although the drug was approved by the FDA, Roche will still have to perform additional studies to confirm the drug works as intended.
It is not yet clear whether the drug will allow people to live longer than traditional methods that are currently in use. But the other checkpoint inhibitors do allow a certain percentage of the population to live for a rather long time, and Roche says this appears to be the case with their drug as well.
Sadly, and unsurprisingly, cost (one of the biggest public and political concerns when it comes to cancer treatment) is high—the drug will set you back $12,500 a month. However, this is roughly in line with the price of the competing checkpoint drugs.