Little Weight, Little Change
From being used in science experiments to weighing everyday objects, the kilogram has been a useful unit of measurement that has remained unchanged for more than a century now. That run could be coming to an end as it and six other base units of measurements are poised to get updated by the International Committee for Weight and Measurements in 2018.

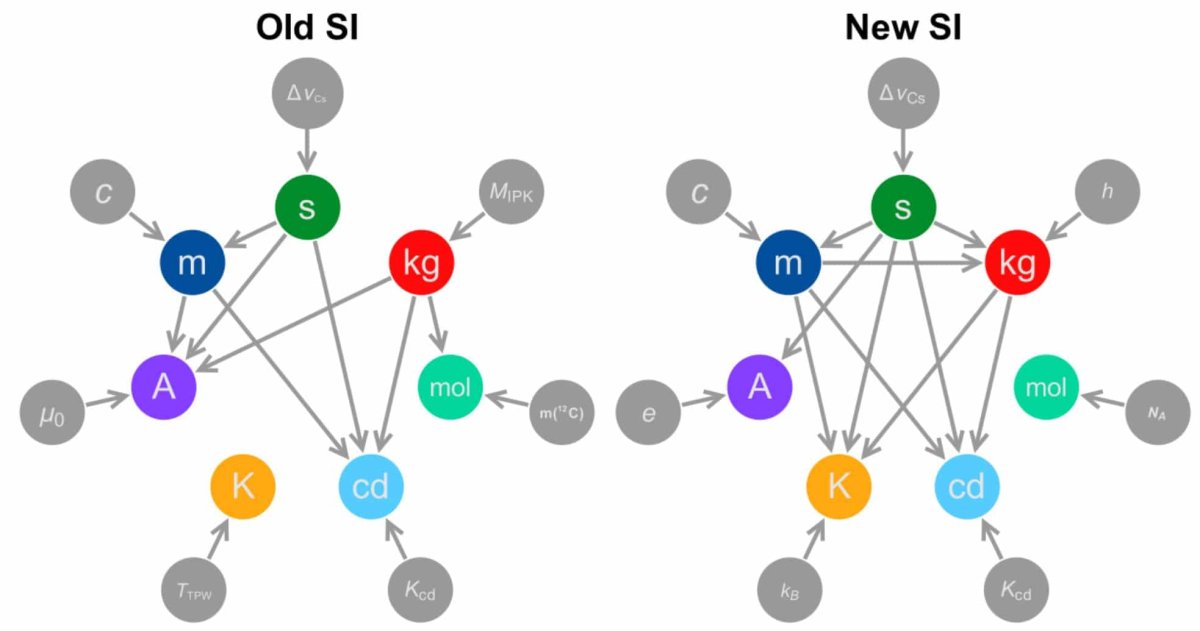
Currently, all seven of the basic units of measurement except the kilogram are based on constant values observed in nature. The kilogram is defined by a physical object called the International Prototype Kilogram. The object, which is kept in a vault in France, has become heavier as of late due to surface contamination. This is a headache for a lot of people, particularly scientists that rely on a constant kilogram for their scientific assumptions.
The new proposal would be to base the kilogram on what is known as Planck’s constant, a value that relates a particle’s energy to its frequency. Scientists have just recently agreed on that value, but they have until July 1, 2017 to publish data that challenges it.
The units ampere, mole, and kelvin are also expected to change. Under the new proposal, the ampere would be based on the elementary charge of an electron, the kelvin would be based on the Boltzmann constant, and the mole would be based on the Avogadro’s constant.
A Smaller Margin For Error
The proposed changes are a result of a better understanding of our universe. As we discover of all these constants in nature, we can push forward with our scientific developments, and existing units of measurement are no longer as accurate as they could be.
While these tiny changes in the metric system are very unlikely to affect the average person, they would give scientists a measuring standard that is as solid a foundation as possible on which to base their research. After all, the fundamental forces of the universe aren’t changing (at least we don’t think they are…).